Echolocation and spatial hearing
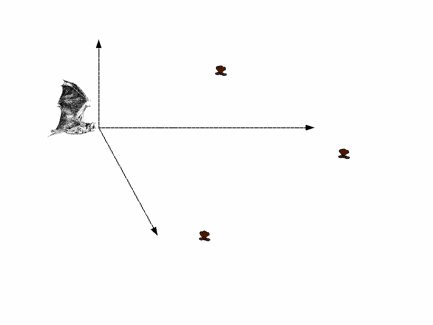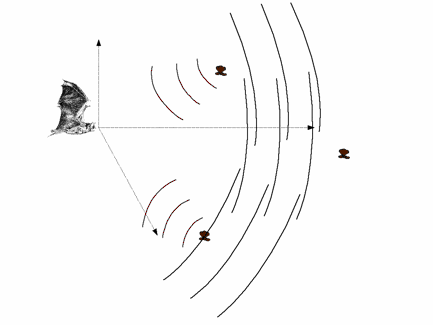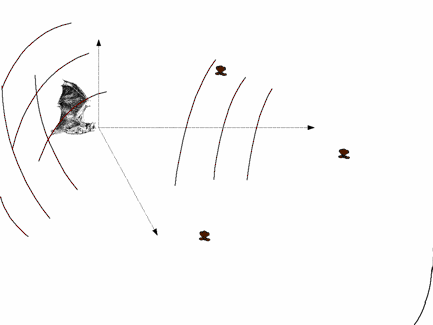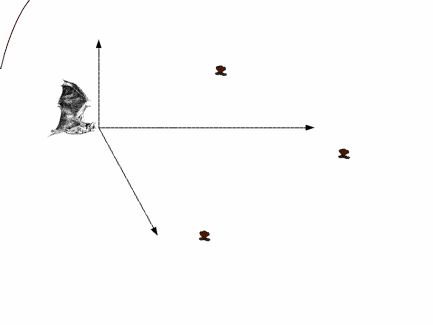
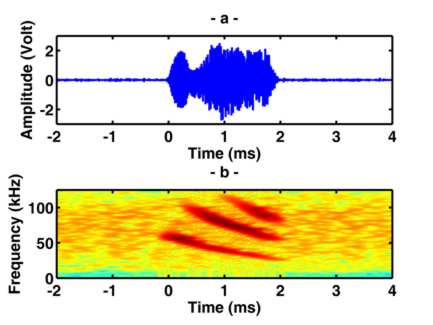
Echolocating bats emit brief ultrasound pulses and listen to the echoes reflected from objects surrounding
them for navigation and prey detection and capture. Bats can determine relative positions of the echo sources
(objects) in terms of distance and direction. Distance is measured as a function of the time elapsed
between the outgoing sound and the incoming echo. To determine direction, bats use the acoustic cues in echo signals
received at each ear. The focus of this paper is on how bats extract
directional properties of echoes.
How do animals localize sound?
Sounds impinging on the ears are transformed by the
shape of the head and ears. External ears, acting like antennas,
amplify or weaken sounds based on their
direction of arrival. These transformations create acoustical cues
that the animal’s brain can use to
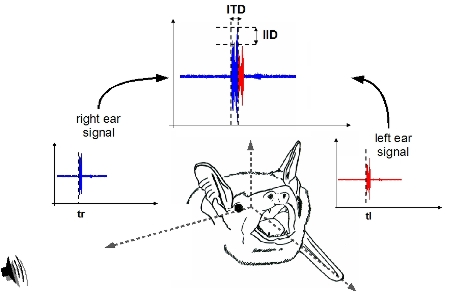
determine sound source location in space. For example: sounds coming
from the right will be received
earlier and with larger intensity at the right ear. These interaural
differences in signal
intensity and arrival time (IID and ITD) decrease as the sound source
moves towards the front and increase
in the opposite direction as it reaches the left side. Therefore,
the auditory system in the brain can use IID and ITD as cues for
horizontal position of the sound source. For echolocating bats IID cues
can also be
used for the localization of the vertical direction –
provided that acoustic signals are wideband [6, 5, 1]. Other direction-dependent features due
to directional modifications can be found in the spectrum of the acoustic signals at each
ears.
Echolocating bats and active sound localization
Bats share similar computational scheme
for passive sound localization with other mammals. But the echoes they detect partly depend on the spectral and
spatial properties of the
echolocation pulses. Bats ensonify the space in front of them to probe
their environment. Acoustic pulses
generated by bats propagate towards different directions with different
intensities. Most bats use echolocation
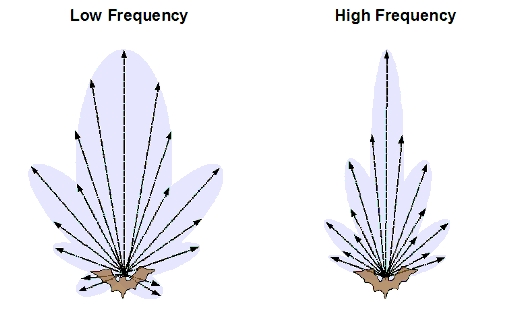
pulses comprised of multiple frequencies. Sound beams generated at high
frequency are directionally
more focused, whereas sound beams generated at low frequencies are
directionally more diffused. Low
frequency sounds are not reflected easily from small objects (like
insects) and travel further. In contrast, high
frequency sounds decay faster yet are easily reflected by the small
objects. Information extracted from echoes containing multiple frequencies provides bats high resolution
representation of their environment. Measurements of sonar emission patterns from unrestrained and freely
vocalizing bats show variation in the echolocation beam shape across
individual vocalizations [3]. Consequently, the bat auditory
system needs to dynamically adapt its computations to accurately localize objects using changing acoustic
inputs.
Statement of the Problem
It is largely accepted that the computation of the
sound localization by the auditory system is based solely on the
acoustic signals received at the ears. Schemes of how the
brain might compute sound location are thus
mostly limited to identifying acoustic cues that map the signal
arriving at the ears to a location in space. Like many other
mammals, bats can move their ears, and therefore acoustic cues for
sound localization even change with ear movements. To accurately
compute the direction of the echo-return, the auditory system
also needs to take ear position into account. Non-auditory signals,
carrying information
about ear position and beam shape, are therefore required for auditory
localization of sound
sources. The auditory system must learn associations between these
extra-auditory signals (e.g. head
and ear position) and the acoustic spatial cues. These demands on the
nervous system suggest
that sound localization is achieved through the interaction of
behavioral control and acoustic
inputs.
Standard approaches that rely on acoustic information alone cannot explain the effects of this
interaction. Moreover, unlike the approach we propose here, by taking an outside observer’s point of view,
they do not address how the auditory system ever acquires the knowledge of the spatial coordinates to
utilize these acoustic cues. We propose a sensorimotor approach to the general problem of sound
localization, with an emphasis on learning that allows spatial information to be acquired and refined by a
naive organism. Thus, the sensorimotor approach does not require prior knowledge of the space by the
system.
A sensorimotor approach for sound localization
Our goal is to show that merely from an
organism’s observations of the sensory consequences of its
self-generated
motions, there is sufficient information to capture the spatial
properties of sounds. The goal is not to find a way
to match acoustic inputs to corresponding spatial parameters but rather
show how the animal could learn the spatial properties of the acoustic
inputs. Normal sensory development requires relevant sensory
inputs
and self-generated actions are also essential for the development of
spatial perception [4] and to adapt
to changes that might occur after its acquisition [7]. Perceptual spatiality of sound sources
is achieved under unnatural acoustic conditions only if a relation between auditory
signals and self-generated motion existed [8].
From an animal’s point of view, the
environment
surrounding the animal is stable as it moves within the environment.
This requires the ability to distinguish sensory input
changes caused by self-generated movements from those that are the
result of changes in the
environment. This can be achieved with the proprioceptive (sense of the
relative positions of the body parts) information, plus the ability to
predict sensory consequences of the organism’s actions, that is,
sensorimotor expertise. We
assert that sensorimotor early experience, during development, is
necessary for accurate sound
localization.
The sensorimotor approach [2]
is based on premises. We first assume
that the organisms are initially unaware of the spatial properties of
the sound sources. Second, we limit the
external sensory information to auditory signals only (e.g. not to
visual input). Third, we postulate an interaction between the
auditory system and the organism’s motor state, that is,
proprioception and motor actions. The first two premises may be viewed
as the worst-case-scenario for sound localization, ignoring any sound
localization
mechanisms that might be hardwired in the brain or aided by vision. But
they don’t significantly constrain
the approach. The third premise, in contrast, is crucial to the
proposed computational
scheme.
Demonstration: Obtaining spatial coordinates by echolocating bats
Our demonstration is a proof of concept. The problem is simplified for the purpose of clarity but does not
effect generality. We make three assumptions:
- We assume that an organism can distinguish the differences between the auditory and
proprioceptive inputs. These inputs can be separated and classified since
auditory inputs can change when the organism is motionless and similarly proprioceptive inputs
can be generated through motion of the body when there is no auditory signal.
- Based on the classification and separability of sensory inputs in these two groups, an organism
can identify the dimension of space through its interaction with the environment [10].
Thus, we also assume that the dimension of auditory space is known.
Auditory space is 2-dimensional since a sound source direction can be
determined by two parameters: horizontal and vertical angles (we ignore
the third dimension of distance to simplify the example).
- We assume that the organism can distinguish the motor actions that can induce a displacement
in the direction of a sound source. An algorithm that demonstrates how a naive system can
identify these special movements is given in [11].
We measured the directional transformations of
sounds due to head and the external ears for big brown
bats. These measurements are used to simulate acoustic signals received
from different directions. Limiting our simulated bat’s movements
to head motions. Simulated sounds with random spectra are
presented from different directions to the artificial bat. When a sound
is present the simulated bat makes three 1o-head movements (up-down, left-to-right, tilt to the right) and records the acoustic inputs
changes in relation to its head movements. These measurements are fed to an unsupervised-learning
algorithm. The algorithm generates a 2-dimensional representation of the data set, a map (Fig.5). Each point on this
map represents a direction in space. Note that our computational method does not use any
a-priori information about the directions of the sounds – only
the measurements taken by the
simulated bat. Learning of this map implies that our simulated bat can
learn the geometric
relations between the points in space. What identifies a point in space
is the unique relationship
between the acoustic input changes and the known set of head movements
that generate them. Auditory input changes caused by a group of head
movements will be identical even if the sound sources themselves are
not.
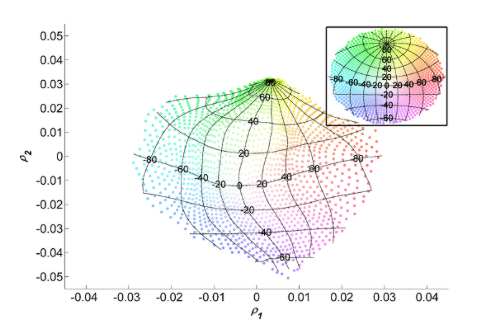
| Figure 5: |
Spatial
locations of the presented sounds are depicted as color coded points in
the stereographic projection of the frontal hemisphere (Inset).
The latitude and longitude lines indicating horizontal and vertical coordinates and the color code allow
comparisons with the map obtained from the unsupervised learning method
and the spatial coordinates of the sound sources. Note that
the points on the map obtained with our computational scheme maintain the same neighborhood
relationships of spatial points in
the frontal space of the bat. |
|
Conclusion
We have proposed a computational method for learning the properties of auditory space using sensorimotor theory
[9], a previously unexplored issue of the problem of sound localization. We have argued that a computational theory
of sound localization should be able to explain the experience-dependent nature of the computation as well
as its dependence on other sensory inputs. The computational method described here provides a framework under which
integration of experience-dependent plasticity and multisensory information processing aspects of sound
localization can be achieved. In conclusion, we have shown that a naive organism can
learn to localize sound based solely on dynamic acoustic inputs and their relation to proprioceptive (motor)
states.
References
[1] M. Aytekin, E. Grassi, M. Sahota, and CF. Moss. The bat head-related transfer function
reveals binaural cues for sound localization in azimuth and elevation. J Acoust Soc Am,
116(6):3594–3605, Dec 2004.
[2] M. Aytekin, J. Z. Simon, and C. F. Moss. A sensorimotor approach to sound localization.
Neural Computation. (In press).
[3] M. Aytekin. Sound Localization by Echolocating Bats. PhD thesis, University of
Maryland, College Park, Maryland, May 2007.
[4] J. Campos, D. Anderson, M. Barbu-Roth, E. Hubbard, M. Hertenstein, and
D. Witherington. Travel broadens the mind. Infancy, 1:149–219, 2000.
[5] Z. M. Fuzessery and G. D. Pollak. Neural mechanisms of sound localization in an
echolocating bat. Science, 225(4663):725–728, Aug 1984.
[6] A. D. Grinnell and V. S. Grinnell. Neural correlates of vertical localization by echo-locating
bats. J Physiol, 181(4):830–851, Dec 1965.
[7] R. Held. Shifts in binaural localization after prolong exposures to atypical
combinations of stimuli. Am J Psychol, 68:526–548, 1955.
[8] J. M. Loomis, C. Hebert, and J. G. Cicinelli. Active localization of virtual sounds. J
Acoust Soc Am, 88(4):1757–64, Oct 1990.
[9] J. K. O’Regan and A No. A sensorimotor account of vision and visual consciousness. Behav
Brain Sci, 24(5):939–73; discussion 973–1031, Oct 2001.
[10] D. Philipona, J. K. O’Regan, and J-P. Nadal. Is there something out there? Inferring space
from sensorimotor dependencies. Neural Comput, 15(9):2029–2049, Sep 2003.
[11] D. Philipona, J. K. O’Regan, J-P Nadal, and O. J.-M.D. Coenen.
Perception of the structure of the physical world using multimodal unknown sensors and
effectors. Advances in Neural Information Processing Systems, 15, 2004.
[ Lay Language Paper Index | Press Room ]