
Figure 1. Picture of an adult gopher tortoise.
[ Lay Language Paper Index | Press Room ]
David Delaney - David.Delaney@erdc.usace.army.mil
Tom Radzio, Joseph Hackler and Andrew Walde of ITS Corporation, Victorville, CA , Matt Hinderliter, of the Nature Conservancy, Camp Shelby, MS
USACERL, P.O. Box 9005
Champaign, IL 61822
Popular version of paper 4aNS5
Presented Friday morning, November 30th, 2007
154th ASA meeting, New Orleans, LA
It is important that natural resource managers on military and non-military lands have access to detailed data on gopher tortoise life history characteristics. Such data are needed to effectively manage and recover gopher tortoise populations, while also providing the information necessary to mitigate for future changes in land management needs. The Department of Defense needs data to understand how gopher tortoise’s (Figure 1) use military lands, to address the question of potential military effects (e.g., training maneuvers, noise) on tortoise populations, while also maintaining the military’s ability to train for national defense. Researchers have commented that military training activities may impact tortoise populations (Berry 1984, USFWS 1994, Wilson et al. 1997), though few researchers have directly studied the effects (e.g., Guyer et al. 1996, Berry et al. 1999, Bowles et al. 1999). The objective of our study is to investigate the potential response of gopher tortoises to human and non-human activities (examples: military training operations, human presence, predators, etc.) in comparison to baseline behavioral patterns to identify potential issues of concern. Our study took place on Camp Shelby, which is a U.S. Army National Guard base in south-central Mississippi, which has a federally threatened population of gopher tortoises.

Figure 1. Picture of an adult gopher tortoise.
Due to the skittish nature of gopher tortoises, and our desire to not impact their behavior while investigating their response to military training operations and other human and non-human activities, we used an automated radio telemetry monitoring system to record tortoise activity levels continuously from March 2006 through September 2007. The basic approach is to record and analyze signals received from radio transmitters mounted on study animals. Because of the directivity pattern of the transmitting antenna on the animal, and because of variations in the tortoises’ juxtaposition to its surroundings, any motion of the animal causes a change in the pulse amplitude, frequency, and polarization of the received signal. Thus, a recording of the temporal variation of the received signal contains considerable information about the motion of the animal, whether it is on the surface or in its burrow. For example, a steady signal recorded at our receiving towers (Figure 2) represent when the tortoise is still (Figure 3: 1st row) or only slightly rotating in its burrow (Figure 3: 2nd row down), while a varying signal (Figure 3: 3rd-5th rows down of stippled areas) indicates when tortoises were active and moving about. These daily activity patterns can be stacked upon one another to develop actograms that illustrate long term patterns of animal activity (Figure 4)

Figure 2. Picture of an automated radio telemetry receiving system.
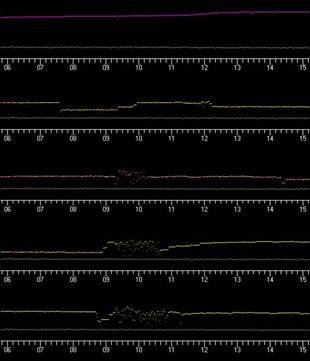
Figure 3. Examples of signal patterns received by automated radio telemetry receiving systems.

Figure 4. Example of a 2 month actogram (rows: midnight to midnight broken into four 6-hr blocks shown by vertical white lines) for a female gopher tortoise from early July through late August 2006. Red illustrate active periods, green show inactive periods for tortoises, and blue represent poor signal data.
To verify data from automated monitoring systems and to quantify and characterize gopher tortoise social behavior and activity, we also video taped a large number of active female tortoises’ at their burrows (Figure 5).

Figure 5. Picture of a gopher tortoise burrow on Camp Shelby, MS. The yellow sign is used to warn soldiers and others to keep away from protected tortoise areas.
Many social interactions between male and female gopher tortoises occur in proximity to female burrows, such as courtship behavior (Figure 6), egg-laying, aggressive interactions between adult tortoises, etc.

Figure 6. Video image of a male gopher tortoise mounting a female attempting to copulate.
Video monitoring systems placed at burrows and along trails/roads also provided information on potential predators (Figure 7), and other types of human activities within study locations (examples: military and non-military vehicles, troop movements, hunters, joggers, etc).

Figure 7. Video imagine of a fox at an active tortoise burrow.
To characterize and quantify military training operations in proximity to tortoise locations, we used professional grade sound recording equipment (Figure 8). This equipment allowed us to measure the number of potential disturbance events, timing of events, sound levels, and frequency spectra of human activities in proximity to tortoise locations.

Figure 8. Photo of the sound recording equipment used on Camp Shelby, MS.
In addition to monitoring tortoise activity, we also collected meteorological data (air and ground temperature, rainfall, etc.) the relative body temperature of tortoises using dataloggers placed on the animals, and temperature down into burrows (Figures 10-12). This allowed us to determine at what temperature tortoises were above and below ground (Figure 9) and how tortoise position varied within burrows depending on the temperature (Figures 10-12).
All equipment was removed from animals at the conclusion of the field study.
Taking all of these different types of technology in concert, we are able to quantify when and for how long tortoises are above and below ground and learn more about what factors influence their behavior. Tortoise temperature and activity levels varied depending on tortoise position above and below ground. Tortoise activity levels and body temperatures were highest while tortoises were away from their burrows, as shown by the 24-hr actogram (red: active/green: inactive).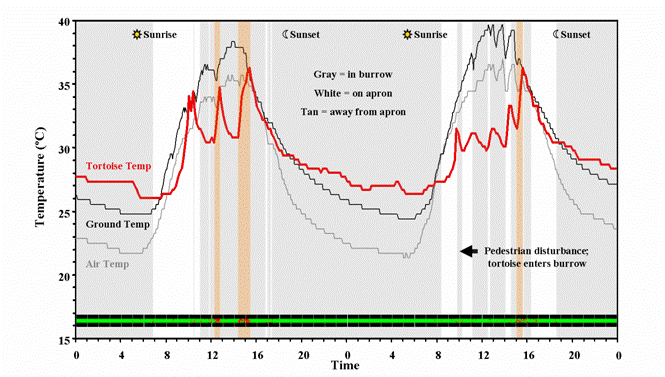
Figure 9. Tortoise temperature and activity is shown in reference to ground/air temperatures, time of day, and human activity.
As expected, we observed that burrow temperature increased with burrow depth (Figures 10-12). Temperature sensors near the burrow entrance had the greatest fluctuation, primarily due to sunlight entering the shallow part of the burrow mouth. Temperatures were much more stable at deeper distances. By comparing tortoise temperatures and burrow temperature at different depths, we are able to estimate how deep tortoises were at different times of day across seasons. Such data helps us better understand tortoise temperature preferences and how tortoises vary their position above and below ground based on temperature, other meteorological conditions, and disturbances by human and non-human activities.
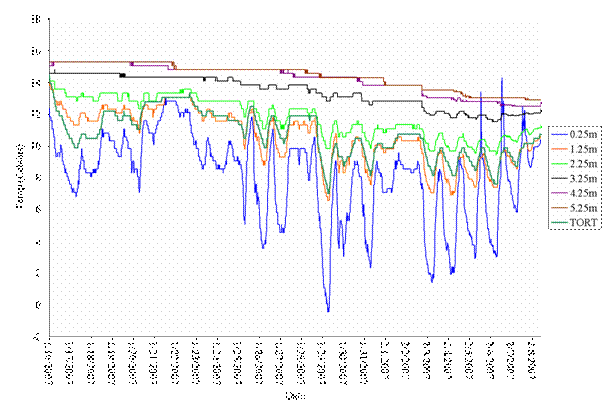
Figure 10. Example of tortoise and burrow temperature data at different depths, showing that the tortoise was between 1.25-2.25 meters deep within its burrow between mid January through early February 2007.
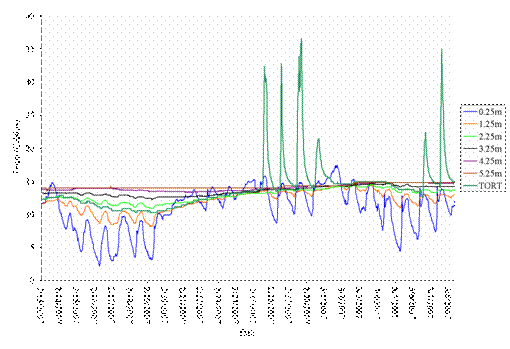
Figure 11. Example of tortoise and burrow temperature data at different depths, showing that the tortoise was between 1.25-2.25 meters deep within its burrow between mid February through late February 2007, but did come above ground for a portion of 6 days between late February through early March 2007.
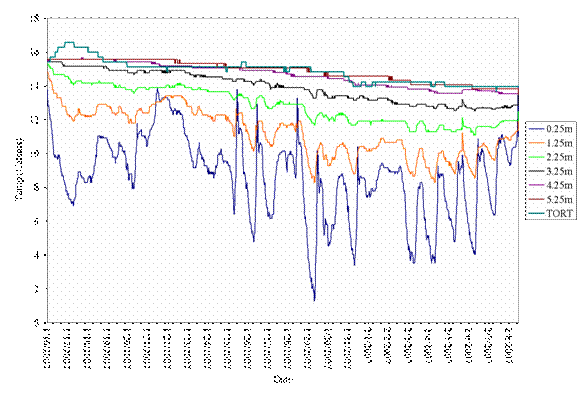
Figure 12. Example of tortoise and burrow temperature data at different depths, showing that the tortoise stays underground between 4.25-5.25 m continuously between mid January through early February 2007.
We are currently analyzing data from this project and therefore cannot discuss our research conclusions on the overall response of gopher tortoises to military training operations or other human and non-human activities. The data examples we have shown do provide some insight in the types of data sets we collected. We believe the data represents a fairly holistic representation of gopher tortoise behavioral patterns which should help resource managers more effectively manage and recover their tortoise populations.
Berry, K.H., G. Goodlett, T. Goodlett, and S. Boland. 1999. Attributes of desert tortoise populations within selected areas at the National Training Center, Fort Irwin, California. Abstract presented at the 24th Meeting and Symposium of the Desert Tortoise Council.
Berry, K. H. and A. P. Woodman. 1984. Methods used in analyzing mortality data for most tortoise populations in California, Nevada, Arizona and Utah. Appendix 7 in K. Berry (ed.). 1984. The status of the gopher tortoise (Gopherus agassizii) in the United States. Gopher Tortoise Council Report to the U.S. Fish and Wildlife Service on Purchase Order No. 11310-0083-81.
Bowles, A.E., S. Eckert, L. Starke, E. Berg, L. Wolski, and J. Matesic, Jr. 1999. Effects
of Flight Noise From Jet Aircraft and Sonic Booms on Hearing, Behavior, Heart Rate, and Oxygen Consumption of Desert Tortoise (Gopherus agassizii). AFRL- HE-WP-TR-1999-0170. Sea World Research Institute, Hubbs Marine Research Center, San Diego, CA. 131 pages.
Guyer, C., K.E. Nicholson, and S. Baucom. 1996. Effects of Tracked Vehicles on Gopher Tortoises (Gopherus polyphemus) at Fort Benning Military Installation, Georgia. Georgia Journal of Science 54:195-203.
USFWS. 1994. Desert Tortoise (Mojave population) recovery plan. USFWS, Portland, OR. 73 pages plus appendices. U.S. Fish and Wildlife Service USFWS. 1987.
Wilson, D.S., H.R. Mushinsky, and R.A. Fischer. 1997. Species Profile: Gopher Tortoise (Gopherus polyphemus) on Military Installations in the Southeastern United States. TR SERDP-97-10. U.S. Army Engineer Waterways Experiment Station, Vicksburg, MS.