Robert E. Apfel -
Robert.Apfel@yale.edu
Department of Mechanical Engineering
Yale University
New Haven, CT 06520-8286
Popular version of paper 2pBB9
Presented Tuesday afternoon, December 5, 2000
ASA/NOISE-CON 2000 Meeting, Newport Beach, CA
Despite the many breakthroughs in cancer drugs, gene treatment, and immunological agents, therapies that actually cure patients have been few and far between. The primary reason for this failure in the midst of so many positive discoveries is that the new agents, which often carry their own toxic side effects, do not get delivered and distributed preferentially in the targeted tissues, sparing normal tissue.
A partnership between researchers at the Yale School of Medicine and Apfel Enterprises, Inc. of New Haven, CT is now examining the feasibility of a novel approach to targeted delivery and distribution that is afforded by suspensions of superheated drops laden with an appropriate therapeutic agent.[i] At body temperature, the drops in such compositions are significantly above the boiling temperature of the liquid composing the drops (that is, they are superheated), but they have not boiled. Each drop, therefore, is like a mousetrap waiting for a triggering event.
Superheated drops compositions have, in the past, been used as detectors for neutrons and other radiations.[ii] Based on the principle of the bubble chamber used in nuclear physics[iii], superheated drops will boil when sufficient energy is deposited in the drops by appropriate radiation-matter interactions.
In the case of superheated drops, the entrained therapeutic agents can be targeted to a particular site by appropriate application of a collimated beam or an implanted source of ionizing radiation or by ultrasound focused on a particular site in a body. (See illustration.) Under appropriate conditions, the superheated drops will be nucleated (triggered), forming bubbles that are about 300 times the volume (or about 6.7 times the diameter) of the original drops. Once delivered, forming bubbles that potentially occlude capillary vessels in the targeted tissue, diagnostic ultrasound can be used to image their presence, as they form good imaging contrast agents.[iv] At higher doses of ultrasound, the bubbles can be induced into motions (pulsation and translation) that help to enhance the diffusion of the therapeutic agent into interstitial tissue, thereby distributing it more effectively.[v]
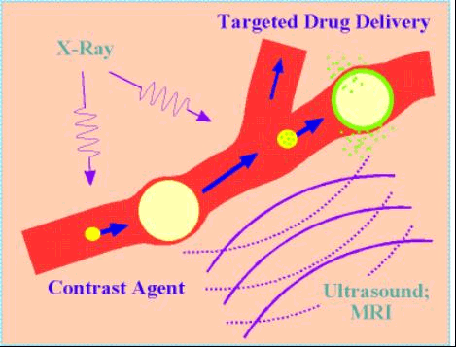
Schematic of targeted drug delivery and contrast agent system. At the lower left a small drop in a capillary vessel is irradiated by either x-rays or ultrasound, causing the bubble to vaporize producing a much larger bubble that can occlude a capillary. That bubble is a good contrast agent for improved ultrasound or MRI imaging. In the upper right a superheated drop is doped with drug. Upon vaporization, a bubble is formed with the drug on the bubble wall. Insonation with higher energy ultrasound will help to drive the drug material into the surrounding tissue.
In one scenario, low dose-rate ionizing irradiation could be used to continually activate droplets passing through a tumor in the circulating blood until the circulating droplets are largely depleted. Such localization of the drug in the area of a tumor would have obvious benefits in chemotherapy and in combined modality chemo-radiation therapy (especially of hard-to-treat solid tumors), as normal tissues outside the irradiated volume could be spared from exposure to the potentially toxic drug, while drug would be selectively delivered to the area of the tumor.[vi],[vii]
The philosophy and the technology needed to produce this therapy delivery, distribution, and diagnostic system would be similar in some ways to those underlying the development of drug carrying liposomes or perfluorochemical emulsions. The therapeutic agent is introduced into the drop liquid under pressure, keeping it in a stable state. This combination is then added to aqueous infusion liquid containing an appropriate encapsulant, and the resultant mixture is agitated by shaking or high-energy ultrasound to emulsify the drug-bearing droplets into the aqueous phase. Once prepared in this manner, the appropriate size distribution of drops can be achieved by allowing the larger drops to be separated by sedimentation or centrifugation. When encapsulated, the drops in the composition will survive the reduction of the pressure to ambient conditions and the passage of the drops through the circulatory system.
At the 140th meeting of the Acoustical Society of America in Newport Beach, California (December 3-8, 2000) measurements are reported that illustrate the methodology of the production of the superheated drop composition. The sensitivity of the drop composition to x-rays and ultrasound is also reported, validating a theoretical approach that allows one to predict whether or not a particular drop material will possess the appropriate sensitivity to radiation and ultrasound. Also, to be sure that the drop composition has the appropriate size distribution, free of larger drops, a new drop analysis system is introduced permitting each sample to be tested and characterized. Probably the biggest challenge in determining the potential for practical application of this system is whether the drop encapsulant is sufficiently strong so as to protect the drops from premature boiling as they make their way to through the circulatory system. Tests are reported that document the behavior of superheated drop compositions in a variety of stress environments. Experiments with rats are also underway to determine toxicity for infused drop compositions without drug present.
-------------------------------------------
[i] U.S. Patent No. 5,840,276 (1998) Activatable infusable dispersions containing drops of a superheated liquid for methods of therapy and diagnosis
[ii] R.E. Apfel U.S. Patent, 4,143,274 (1979), and R.E.Apfel, "The Superheated Drop Detector," Nuclear Instrum. and Meth. 162, 603-608 (1979).
[iii] D.Glaser, Phys. Review, 87, 665(1952).
[iv] L Needleman and F. Forsberg, "Contrast agents in ultrasound," Ultrasound Quarterly 13, 121-138(1996).
[v] C.W. Francis and P. N. Riggs, "Acceleration of thrombolysis by ultrasound in two rabbit models," 99, 2466(abstract)(1996).
[vi] S. Rockwell and A.C. Sartorelli, "Interactions between mitomycin C and radiation" Antitumor Drug-Radiation Interactions, in B.T. Hill and A.S. Bellamy eds. CRC Press, 1990, pp.125-140.
[vii] B. G. Haffty, Y.H. Son, C.T. Sasaki, R. Papac, D. Fischer,S. Rockwell, A.C. Sartorelli, and J.J. Fischer,"Mitomycin C as an adjunct to postoperative radiation therapy in squamous cell carcinoma of the head and the neck: results from two randomized clinical tirals," Int. Jour. Rad. Oncol. Bio. Phys. ,27,241-250(1993).